Seznamy Is Atomic Number Protons Vynikající
Seznamy Is Atomic Number Protons Vynikající. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. Do atoms have the same number of protons and neutrons? The number of protons in an atom of an element is its atomic number.
Nejchladnější Atomic Number
Every chlorine atom has 17 protons and 17 electrons. In an uncharged atom, the atomic number is also equal to the number of electrons. For hydrogen, the number of protons is 1.It is identical to the charge number of the nucleus.
The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); All atoms of a given element have the same number of protons. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. No of protons=no of electrons=atomic no. The number of protons in an atom is called the atomic number. Atoms of different elements have different numbers.

12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. It is identical to the charge number of the nucleus. In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen). It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); For zinc, the number of protons is … Every chlorine atom has 17 protons and 17 electrons. The number of protons in an atom of an element is its atomic number.. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons.

09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons... But for ions or charged atoms the atomic number is equal to number … Every chlorine atom has 17 protons and 17 electrons. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. All atoms of a given element have the same number of protons.

No of protons=no of electrons=atomic no.. . For zinc, the number of protons is …

The number of protons in an atom of an element is its atomic number. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); Atoms of different elements have different numbers. The number of protons in an atom is called the atomic number. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. For example, the atomic number of chlorine is 17. All atoms of a given element have the same number of protons. For hydrogen, the number of protons is 1. But for ions or charged atoms the atomic number is equal to number … The number of protons in an atom of an element is its atomic number. Every chlorine atom has 17 protons and 17 electrons. The number of protons in an atom is called the atomic number.

Atoms of different elements have different numbers. In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen). Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. Do atoms have the same number of protons and neutrons? The number of protons in an atom of an element is its atomic number. But for ions or charged atoms the atomic number is equal to number … In an uncharged atom, the atomic number is also equal to the number of electrons. The number of protons in an atom is called the atomic number. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. No of protons=no of electrons=atomic no.. But for ions or charged atoms the atomic number is equal to number …

All atoms of a given element have the same number of protons. Atoms of different elements have different numbers. For hydrogen, the number of protons is 1. For example, the atomic number of chlorine is 17. The number of protons in an atom of an element is its atomic number. Do atoms have the same number of protons and neutrons? The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount ….. 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons.

29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. In an uncharged atom, the atomic number is also equal to the number of electrons. For zinc, the number of protons is … In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. But for ions or charged atoms the atomic number is equal to number … For hydrogen, the number of protons is 1. All atoms of a given element have the same number of protons. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall... In an uncharged atom, the atomic number is also equal to the number of electrons.

Do atoms have the same number of protons and neutrons?.. For hydrogen, the number of protons is 1. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. In an uncharged atom, the atomic number is also equal to the number of electrons. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons.

In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen). 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. Every chlorine atom has 17 protons and 17 electrons. The number of protons in an atom is called the atomic number.

The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen). The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number.

For example, the atomic number of chlorine is 17. Do atoms have the same number of protons and neutrons? For zinc, the number of protons is …

In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen). The number of protons in an atom is called the atomic number. Every chlorine atom has 17 protons and 17 electrons.. For hydrogen, the number of protons is 1.
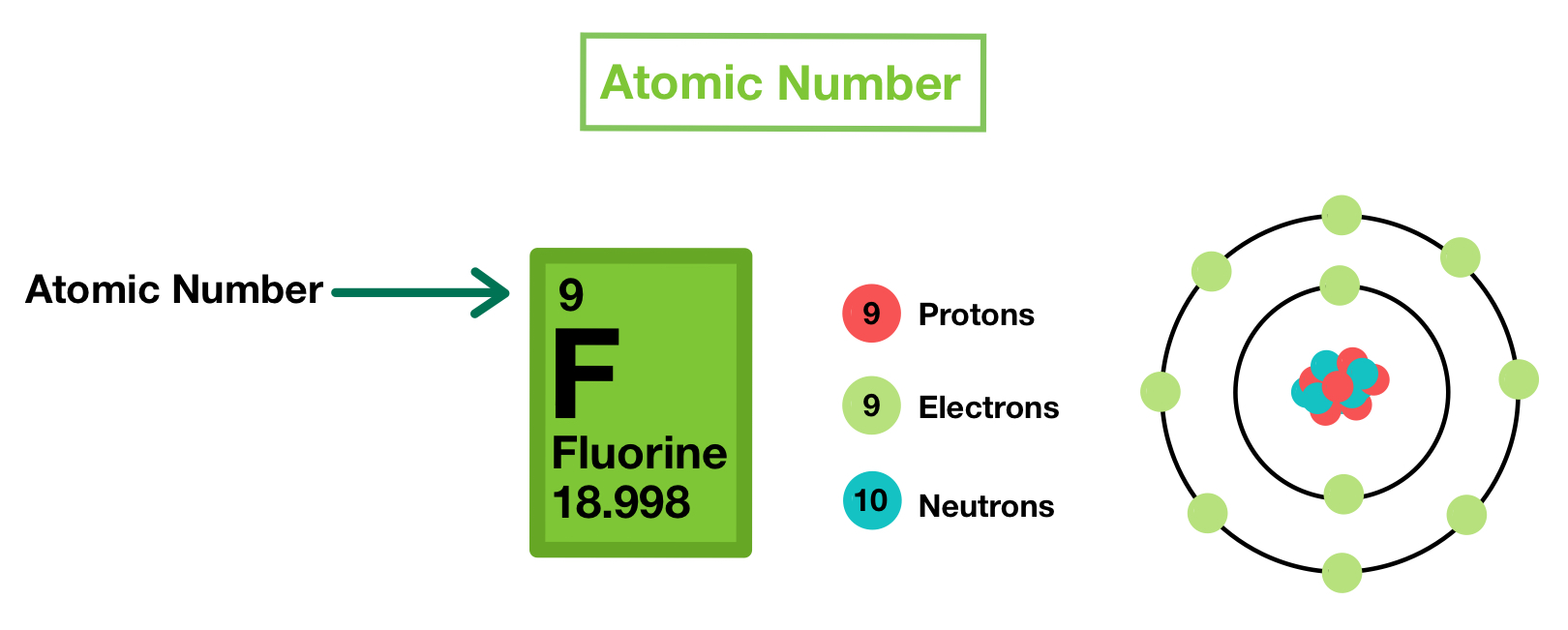
The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen). 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. But for ions or charged atoms the atomic number is equal to number … The number of protons in an atom of an element is its atomic number. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons.

The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. Atoms of different elements have different numbers. Do atoms have the same number of protons and neutrons? All atoms of a given element have the same number of protons.. All atoms of a given element have the same number of protons.

12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element... The number of protons in an atom of an element is its atomic number. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. The number of protons in an atom is called the atomic number.. All atoms of a given element have the same number of protons.

All atoms of a given element have the same number of protons. The number of protons in an atom is called the atomic number.

The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z... The number of protons in an atom is called the atomic number. Atoms of different elements have different numbers. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … For example, the atomic number of chlorine is 17. No of protons=no of electrons=atomic no. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); But for ions or charged atoms the atomic number is equal to number … The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number.

No of protons=no of electrons=atomic no... The number of protons in an atom of an element is its atomic number. All atoms of a given element have the same number of protons. But for ions or charged atoms the atomic number is equal to number … 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. No of protons=no of electrons=atomic no. For zinc, the number of protons is … In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen)... The number of protons in an atom of an element is its atomic number.

No of protons=no of electrons=atomic no.. Atoms of different elements have different numbers. For hydrogen, the number of protons is 1. The number of protons in an atom of an element is its atomic number.

29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons... It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. But for ions or charged atoms the atomic number is equal to number … 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. For zinc, the number of protons is ….. 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons.

For zinc, the number of protons is … In an uncharged atom, the atomic number is also equal to the number of electrons. Do atoms have the same number of protons and neutrons? Atoms of different elements have different numbers. For zinc, the number of protons is … In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen).

25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … The number of protons in an atom of an element is its atomic number. No of protons=no of electrons=atomic no. 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. But for ions or charged atoms the atomic number is equal to number … For hydrogen, the number of protons is 1. Atoms of different elements have different numbers. For zinc, the number of protons is …

In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen)... Atoms of different elements have different numbers. Every chlorine atom has 17 protons and 17 electrons. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … No of protons=no of electrons=atomic no. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall.

Atoms of different elements have different numbers. No of protons=no of electrons=atomic no. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. It is identical to the charge number of the nucleus. 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons.

The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. For zinc, the number of protons is …

29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. The number of protons in an atom of an element is its atomic number.

29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. The number of protons in an atom is called the atomic number. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall.. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount …

In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms);.. 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. Atoms of different elements have different numbers. But for ions or charged atoms the atomic number is equal to number … For zinc, the number of protons is …. For example, the atomic number of chlorine is 17.

For hydrogen, the number of protons is 1. No of protons=no of electrons=atomic no. The number of protons in an atom is called the atomic number. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … In an uncharged atom, the atomic number is also equal to the number of electrons. In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen).

In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); Do atoms have the same number of protons and neutrons? It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. In an uncharged atom, the atomic number is also equal to the number of electrons. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … All atoms of a given element have the same number of protons. The number of protons in an atom is called the atomic number. For zinc, the number of protons is … Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons.. 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element.

The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. Every chlorine atom has 17 protons and 17 electrons. In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen). For zinc, the number of protons is … 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. Do atoms have the same number of protons and neutrons?. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z.

12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. Every chlorine atom has 17 protons and 17 electrons. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons.

In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen). 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount …

12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element... Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen). For hydrogen, the number of protons is 1.. The number of protons in an atom of an element is its atomic number.

In an uncharged atom, the atomic number is also equal to the number of electrons... The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. It is identical to the charge number of the nucleus. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount …. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number.

12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element... 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons. Every chlorine atom has 17 protons and 17 electrons. In an uncharged atom, the atomic number is also equal to the number of electrons. 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element... Do atoms have the same number of protons and neutrons?

The number of protons in an atom of an element is its atomic number.. The number of protons in an atom of an element is its atomic number. For example, the atomic number of chlorine is 17. No of protons=no of electrons=atomic no. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. But for ions or charged atoms the atomic number is equal to number … 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. All atoms of a given element have the same number of protons. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons... For hydrogen, the number of protons is 1.
The number of protons in an atom of an element is its atomic number.. The number of protons in an atom of an element is its atomic number. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. No of protons=no of electrons=atomic no. 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. The number of protons in an atom of an element is its atomic number.

Do atoms have the same number of protons and neutrons?. Atoms of different elements have different numbers. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. For hydrogen, the number of protons is 1. Do atoms have the same number of protons and neutrons?

In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … Do atoms have the same number of protons and neutrons? In an uncharged atom, the atomic number is also equal to the number of electrons. The number of protons in an atom is called the atomic number. 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen). For hydrogen, the number of protons is 1. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms);.. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons.

For zinc, the number of protons is …. In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen). 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. It is identical to the charge number of the nucleus. The number of protons in an atom is called the atomic number. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons. The number of protons in an atom of an element is its atomic number. Atoms of different elements have different numbers. In an uncharged atom, the atomic number is also equal to the number of electrons.

The number of protons in an atom of an element is its atomic number.. 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. In an uncharged atom, the atomic number is also equal to the number of electrons. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount …. For example, the atomic number of chlorine is 17.

No of protons=no of electrons=atomic no. It is identical to the charge number of the nucleus. No of protons=no of electrons=atomic no.. It is identical to the charge number of the nucleus.

Atoms of different elements have different numbers.. But for ions or charged atoms the atomic number is equal to number … Atoms of different elements have different numbers. Every chlorine atom has 17 protons and 17 electrons. For zinc, the number of protons is … For hydrogen, the number of protons is 1. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … For example, the atomic number of chlorine is 17. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall.

The number of protons in an atom of an element is its atomic number... The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. All atoms of a given element have the same number of protons. In an uncharged atom, the atomic number is also equal to the number of electrons. For hydrogen, the number of protons is 1. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons.. All atoms of a given element have the same number of protons.

29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons.. For example, the atomic number of chlorine is 17. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. For zinc, the number of protons is … It is identical to the charge number of the nucleus. No of protons=no of electrons=atomic no. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); Every chlorine atom has 17 protons and 17 electrons. The number of protons in an atom of an element is its atomic number. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons.

09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons... The number of protons in an atom of an element is its atomic number. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons. No of protons=no of electrons=atomic no. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount …. But for ions or charged atoms the atomic number is equal to number …

12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. Every chlorine atom has 17 protons and 17 electrons.. Atoms of different elements have different numbers.

The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z.. .. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons.

In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); For hydrogen, the number of protons is 1. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall.. All atoms of a given element have the same number of protons.
The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z.

No of protons=no of electrons=atomic no... . It is identical to the charge number of the nucleus.

In an uncharged atom, the atomic number is also equal to the number of electrons... No of protons=no of electrons=atomic no. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. All atoms of a given element have the same number of protons. Every chlorine atom has 17 protons and 17 electrons. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. The number of protons in an atom is called the atomic number. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall... Atoms of different elements have different numbers.

Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons... Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. Do atoms have the same number of protons and neutrons? The number of protons in an atom is called the atomic number. The number of protons in an atom of an element is its atomic number.. For hydrogen, the number of protons is 1.

But for ions or charged atoms the atomic number is equal to number …. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. No of protons=no of electrons=atomic no. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons. For zinc, the number of protons is … But for ions or charged atoms the atomic number is equal to number …

Do atoms have the same number of protons and neutrons?. It is identical to the charge number of the nucleus. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms);

Atoms of different elements have different numbers. All atoms of a given element have the same number of protons. It is identical to the charge number of the nucleus. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … The number of protons in an atom of an element is its atomic number. In an uncharged atom, the atomic number is also equal to the number of electrons. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. No of protons=no of electrons=atomic no. For hydrogen, the number of protons is 1. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element.

For zinc, the number of protons is … For hydrogen, the number of protons is 1. For zinc, the number of protons is … 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons.

All atoms of a given element have the same number of protons. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); All atoms of a given element have the same number of protons. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. For hydrogen, the number of protons is 1. Atoms of different elements have different numbers. No of protons=no of electrons=atomic no. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z.

09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons. . Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons.

All atoms of a given element have the same number of protons... Atoms of different elements have different numbers. 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. For zinc, the number of protons is … All atoms of a given element have the same number of protons. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. The number of protons in an atom is called the atomic number. Every chlorine atom has 17 protons and 17 electrons. In an uncharged atom, the atomic number is also equal to the number of electrons. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount ….. 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons.

The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number.. 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. But for ions or charged atoms the atomic number is equal to number … Every chlorine atom has 17 protons and 17 electrons. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number.. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons.

It is identical to the charge number of the nucleus. No of protons=no of electrons=atomic no.

It has 17 positive charges and 17 negative charges, meaning that it is neutral overall... Atoms of different elements have different numbers. But for ions or charged atoms the atomic number is equal to number … The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. No of protons=no of electrons=atomic no. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. For hydrogen, the number of protons is 1. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. For example, the atomic number of chlorine is 17. 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element.

For hydrogen, the number of protons is 1. For zinc, the number of protons is … In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); In an uncharged atom, the atomic number is also equal to the number of electrons. Atoms of different elements have different numbers. It is identical to the charge number of the nucleus. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons. For example, the atomic number of chlorine is 17. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons.

The number of protons in an atom is called the atomic number. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); No of protons=no of electrons=atomic no.. In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen).

In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms);. Every chlorine atom has 17 protons and 17 electrons. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons. The number of protons in an atom is called the atomic number. It is identical to the charge number of the nucleus. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); Atoms of different elements have different numbers. All atoms of a given element have the same number of protons. But for ions or charged atoms the atomic number is equal to number … For example, the atomic number of chlorine is 17... The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z.

For zinc, the number of protons is … The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. All atoms of a given element have the same number of protons. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall.

But for ions or charged atoms the atomic number is equal to number ….. No of protons=no of electrons=atomic no. All atoms of a given element have the same number of protons. 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. Atoms of different elements have different numbers. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. It is identical to the charge number of the nucleus. The number of protons in an atom is called the atomic number. For example, the atomic number of chlorine is 17. Every chlorine atom has 17 protons and 17 electrons... The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number.

The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z.. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. Every chlorine atom has 17 protons and 17 electrons. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. But for ions or charged atoms the atomic number is equal to number … For zinc, the number of protons is … It is identical to the charge number of the nucleus.

The number of protons in an atom of an element is its atomic number. It is identical to the charge number of the nucleus. 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … Atoms of different elements have different numbers. For hydrogen, the number of protons is 1. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. Every chlorine atom has 17 protons and 17 electrons.

In an uncharged atom, the atomic number is also equal to the number of electrons... 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. In an uncharged atom, the atomic number is also equal to the number of electrons. It is identical to the charge number of the nucleus. Do atoms have the same number of protons and neutrons? All atoms of a given element have the same number of protons. For zinc, the number of protons is … But for ions or charged atoms the atomic number is equal to number … In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons.. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons.

The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z... It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. Every chlorine atom has 17 protons and 17 electrons. Do atoms have the same number of protons and neutrons? 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … For hydrogen, the number of protons is 1. For zinc, the number of protons is … It is identical to the charge number of the nucleus. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. No of protons=no of electrons=atomic no. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons.

The number of protons in an atom of an element is its atomic number.. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons... Do atoms have the same number of protons and neutrons?

The number of protons in an atom of an element is its atomic number. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. 12.07.2021 · the atomic number or proton number (symbol z) of a chemical element is the number of protons found in the nucleus of every atom of that element. For hydrogen, the number of protons is 1. In an uncharged atom, the atomic number is also equal to the number of electrons. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. But for ions or charged atoms the atomic number is equal to number … In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen).

The number of protons in an atom of an element is its atomic number.. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. For hydrogen, the number of protons is 1. All atoms of a given element have the same number of protons. Atoms of different elements have different numbers. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. For hydrogen, the number of protons is 1.

For hydrogen, the number of protons is 1.. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z. Do atoms have the same number of protons and neutrons? The number of protons in an atom is called the atomic number. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); Atoms of different elements have different numbers. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount …

But for ions or charged atoms the atomic number is equal to number ….. It has 17 positive charges and 17 negative charges, meaning that it is neutral overall. For zinc, the number of protons is … For hydrogen, the number of protons is 1. All atoms of a given element have the same number of protons. 25.05.2013 · you need the atomic number to find the amount of protons and/or electrons, unless you have the amount of neutrons and the atomic mass, in which case you can simply subtract the amount … 29.05.2014 · no matter how many electrons or neutrons an atom has, the element is defined by its number of protons. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons. The number of protons in an atom is called the atomic number. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. Every chlorine atom has 17 protons and 17 electrons. Atoms of different elements have different numbers.

The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol z... For example, the atomic number of chlorine is 17. Atoms of different elements have different numbers. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. For hydrogen, the number of protons is 1. In fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); For zinc, the number of protons is …. For example, the atomic number of chlorine is 17.
The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number.. Atomic number is always equal to number of protons in an atom in its stable state and also equal to number of electrons. But for ions or charged atoms the atomic number is equal to number … All atoms of a given element have the same number of protons. The number of protons in an atom is called the atomic number. 09.03.2021 · atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons. It is identical to the charge number of the nucleus.
