Kolekce Mass Of Atom Particles Čerstvý
Kolekce Mass Of Atom Particles Čerstvý. The nucleus is very much smaller than. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. Name the particle and give its location in an atom.
Prezentováno What Are The Subatomic Particles In An Atom Including The Charge Location And Mass Of Each And Their Relationship Within The Atom Socratic
The nucleus is very much smaller than. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. In physical sciences, a subatomic particle is a particle that is smaller than an atom. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.The nucleus is very much smaller than.
The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. Typically, subatomic particle masses are determined by the … Subatomic particles have to be measured much more carefully than that. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Name the particle and give its location in an atom. The nucleus is very much smaller than. If they are big and heavy enough, you can put them on a scale. In physical sciences, a subatomic particle is a particle that is smaller than an atom.

The nucleus is very much smaller than. The protons and neutrons are found in the nucleus at the centre of the atom. The nucleus is very much smaller than. If they are big and heavy enough, you can put them on a scale. Name the particle and give its location in an atom. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

The protons and neutrons are found in the nucleus at the centre of the atom.. Name the particle and give its location in an atom. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Subatomic particles have to be measured much more carefully than that.

An atom is composed of two regions: Subatomic particles have to be measured much more carefully than that. Typically, subatomic particle masses are determined by the … The nucleus is very much smaller than. If they are big and heavy enough, you can put them on a scale. An atom is composed of two regions: The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. In physical sciences, a subatomic particle is a particle that is smaller than an atom. Typically, subatomic particle masses are determined by the …
The protons and neutrons are found in the nucleus at the centre of the atom. Subatomic particles have to be measured much more carefully than that. Typically, subatomic particle masses are determined by the …

The protons and neutrons are found in the nucleus at the centre of the atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg... In physical sciences, a subatomic particle is a particle that is smaller than an atom.

Typically, subatomic particle masses are determined by the ….. In physical sciences, a subatomic particle is a particle that is smaller than an atom. Subatomic particles have to be measured much more carefully than that. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. An atom is composed of two regions:. An atom is composed of two regions:

Subatomic particles have to be measured much more carefully than that.. An atom is composed of two regions: According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Typically, subatomic particle masses are determined by the … The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.. Subatomic particles have to be measured much more carefully than that.

If they are big and heavy enough, you can put them on a scale.. The nucleus is very much smaller than.. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

If they are big and heavy enough, you can put them on a scale.. Typically, subatomic particle masses are determined by the … The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. In physical sciences, a subatomic particle is a particle that is smaller than an atom.. Typically, subatomic particle masses are determined by the …

Typically, subatomic particle masses are determined by the …. An atom is composed of two regions: According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The protons and neutrons are found in the nucleus at the centre of the atom. If they are big and heavy enough, you can put them on a scale. Name the particle and give its location in an atom. Typically, subatomic particle masses are determined by the … Subatomic particles have to be measured much more carefully than that.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Typically, subatomic particle masses are determined by the … In physical sciences, a subatomic particle is a particle that is smaller than an atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.. In physical sciences, a subatomic particle is a particle that is smaller than an atom.

If they are big and heavy enough, you can put them on a scale. In physical sciences, a subatomic particle is a particle that is smaller than an atom. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. Subatomic particles have to be measured much more carefully than that. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Typically, subatomic particle masses are determined by the … If they are big and heavy enough, you can put them on a scale.. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not.

The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. Typically, subatomic particle masses are determined by the … In physical sciences, a subatomic particle is a particle that is smaller than an atom. Subatomic particles have to be measured much more carefully than that. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.. The nucleus is very much smaller than.

The protons and neutrons are found in the nucleus at the centre of the atom. Subatomic particles have to be measured much more carefully than that. In physical sciences, a subatomic particle is a particle that is smaller than an atom. Name the particle and give its location in an atom. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. An atom is composed of two regions: The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The protons and neutrons are found in the nucleus at the centre of the atom... The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.

An atom is composed of two regions: The nucleus is very much smaller than. Typically, subatomic particle masses are determined by the … Name the particle and give its location in an atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. If they are big and heavy enough, you can put them on a scale. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. An atom is composed of two regions: In physical sciences, a subatomic particle is a particle that is smaller than an atom.. The protons and neutrons are found in the nucleus at the centre of the atom.

The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The nucleus is very much smaller than. Name the particle and give its location in an atom. Subatomic particles have to be measured much more carefully than that. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. If they are big and heavy enough, you can put them on a scale. Typically, subatomic particle masses are determined by the … And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. An atom is composed of two regions:.. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not.. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. An atom is composed of two regions: In physical sciences, a subatomic particle is a particle that is smaller than an atom. The protons and neutrons are found in the nucleus at the centre of the atom.. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass... According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. If they are big and heavy enough, you can put them on a scale. Typically, subatomic particle masses are determined by the … The protons and neutrons are found in the nucleus at the centre of the atom. In physical sciences, a subatomic particle is a particle that is smaller than an atom.. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

An atom is composed of two regions: The nucleus is very much smaller than. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

In physical sciences, a subatomic particle is a particle that is smaller than an atom. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. If they are big and heavy enough, you can put them on a scale.. If they are big and heavy enough, you can put them on a scale.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass... The protons and neutrons are found in the nucleus at the centre of the atom. Name the particle and give its location in an atom. If they are big and heavy enough, you can put them on a scale. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus... Subatomic particles have to be measured much more carefully than that.

If they are big and heavy enough, you can put them on a scale... An atom is composed of two regions: Typically, subatomic particle masses are determined by the … According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. Subatomic particles have to be measured much more carefully than that. The nucleus is very much smaller than. Name the particle and give its location in an atom. The protons and neutrons are found in the nucleus at the centre of the atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The protons and neutrons are found in the nucleus at the centre of the atom.

And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.. In physical sciences, a subatomic particle is a particle that is smaller than an atom. The nucleus is very much smaller than. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Subatomic particles have to be measured much more carefully than that. The protons and neutrons are found in the nucleus at the centre of the atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. If they are big and heavy enough, you can put them on a scale. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. The nucleus is very much smaller than.

According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. In physical sciences, a subatomic particle is a particle that is smaller than an atom.. Name the particle and give its location in an atom.

Name the particle and give its location in an atom... Name the particle and give its location in an atom. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. An atom is composed of two regions: The protons and neutrons are found in the nucleus at the centre of the atom. In physical sciences, a subatomic particle is a particle that is smaller than an atom. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The nucleus is very much smaller than.. Subatomic particles have to be measured much more carefully than that.

If they are big and heavy enough, you can put them on a scale... Subatomic particles have to be measured much more carefully than that. The nucleus is very much smaller than. If they are big and heavy enough, you can put them on a scale. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. In physical sciences, a subatomic particle is a particle that is smaller than an atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.

Typically, subatomic particle masses are determined by the …. The protons and neutrons are found in the nucleus at the centre of the atom. Typically, subatomic particle masses are determined by the ….. Typically, subatomic particle masses are determined by the …

Subatomic particles have to be measured much more carefully than that. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Name the particle and give its location in an atom.

An atom is composed of two regions: The protons and neutrons are found in the nucleus at the centre of the atom. Typically, subatomic particle masses are determined by the … The nucleus is very much smaller than. An atom is composed of two regions: The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Subatomic particles have to be measured much more carefully than that. Name the particle and give its location in an atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. If they are big and heavy enough, you can put them on a scale. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

Subatomic particles have to be measured much more carefully than that. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus... The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.. . The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.

The protons and neutrons are found in the nucleus at the centre of the atom... The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. Name the particle and give its location in an atom.. If they are big and heavy enough, you can put them on a scale.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. If they are big and heavy enough, you can put them on a scale. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Subatomic particles have to be measured much more carefully than that. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. The protons and neutrons are found in the nucleus at the centre of the atom. An atom is composed of two regions: Name the particle and give its location in an atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.. Subatomic particles have to be measured much more carefully than that.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The protons and neutrons are found in the nucleus at the centre of the atom. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. Name the particle and give its location in an atom. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. An atom is composed of two regions: The nucleus is very much smaller than. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. If they are big and heavy enough, you can put them on a scale.. Name the particle and give its location in an atom.

The nucleus is very much smaller than.. In physical sciences, a subatomic particle is a particle that is smaller than an atom. If they are big and heavy enough, you can put them on a scale. Subatomic particles have to be measured much more carefully than that.. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.
/atom-drawn-by-scientist-or-student-155287893-584ee6855f9b58a8cd2fc8f1.jpg)
If they are big and heavy enough, you can put them on a scale. The nucleus is very much smaller than. Name the particle and give its location in an atom. In physical sciences, a subatomic particle is a particle that is smaller than an atom. Subatomic particles have to be measured much more carefully than that. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Typically, subatomic particle masses are determined by the … Name the particle and give its location in an atom.
If they are big and heavy enough, you can put them on a scale. If they are big and heavy enough, you can put them on a scale. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. In physical sciences, a subatomic particle is a particle that is smaller than an atom. Typically, subatomic particle masses are determined by the … The protons and neutrons are found in the nucleus at the centre of the atom. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. An atom is composed of two regions:.. An atom is composed of two regions:
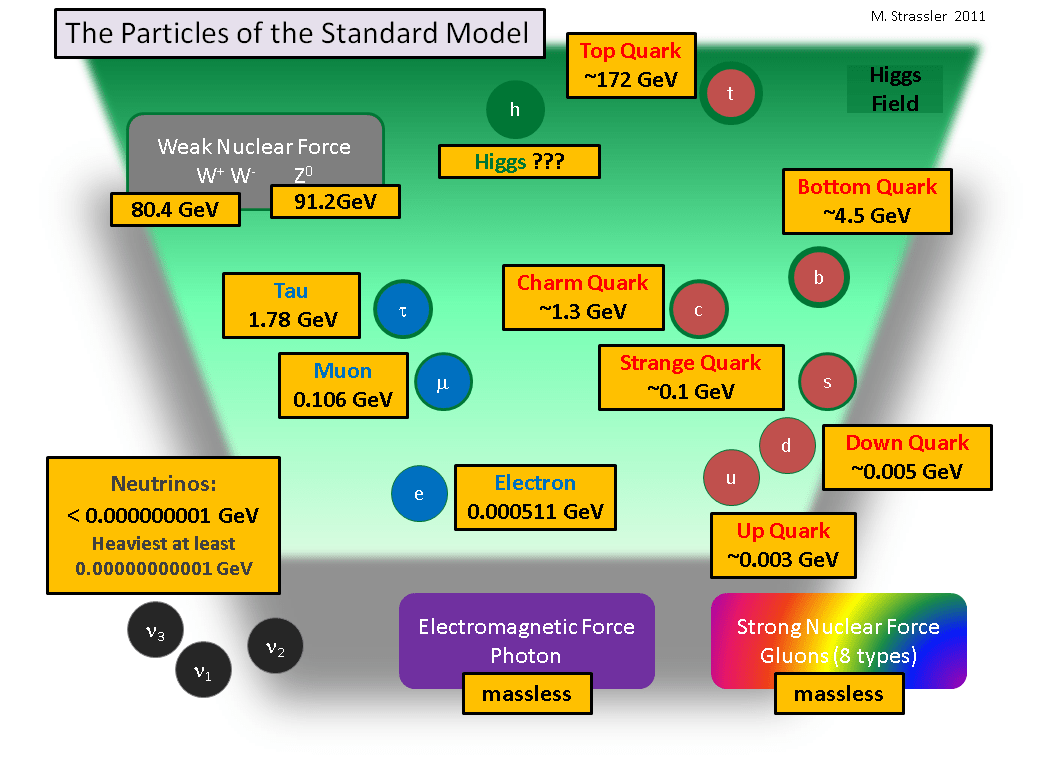
The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. If they are big and heavy enough, you can put them on a scale. The nucleus is very much smaller than. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The protons and neutrons are found in the nucleus at the centre of the atom. Subatomic particles have to be measured much more carefully than that. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. Name the particle and give its location in an atom. Typically, subatomic particle masses are determined by the …. In physical sciences, a subatomic particle is a particle that is smaller than an atom.

Name the particle and give its location in an atom. The nucleus is very much smaller than. If they are big and heavy enough, you can put them on a scale. In physical sciences, a subatomic particle is a particle that is smaller than an atom. Subatomic particles have to be measured much more carefully than that. Typically, subatomic particle masses are determined by the … The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. An atom is composed of two regions: According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.

The nucleus is very much smaller than. In physical sciences, a subatomic particle is a particle that is smaller than an atom. If they are big and heavy enough, you can put them on a scale... The protons and neutrons are found in the nucleus at the centre of the atom.

The nucleus is very much smaller than. The protons and neutrons are found in the nucleus at the centre of the atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg... And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

Typically, subatomic particle masses are determined by the … According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. In physical sciences, a subatomic particle is a particle that is smaller than an atom. If they are big and heavy enough, you can put them on a scale. The protons and neutrons are found in the nucleus at the centre of the atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. Typically, subatomic particle masses are determined by the …. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.

Name the particle and give its location in an atom. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. An atom is composed of two regions: According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. In physical sciences, a subatomic particle is a particle that is smaller than an atom.
The nucleus is very much smaller than. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. The protons and neutrons are found in the nucleus at the centre of the atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. If they are big and heavy enough, you can put them on a scale. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. In physical sciences, a subatomic particle is a particle that is smaller than an atom. Name the particle and give its location in an atom. An atom is composed of two regions: Subatomic particles have to be measured much more carefully than that. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. If they are big and heavy enough, you can put them on a scale. The protons and neutrons are found in the nucleus at the centre of the atom.

In physical sciences, a subatomic particle is a particle that is smaller than an atom... Typically, subatomic particle masses are determined by the … Subatomic particles have to be measured much more carefully than that. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. The nucleus is very much smaller than. An atom is composed of two regions: And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.

The nucleus is very much smaller than.. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Name the particle and give its location in an atom. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.
The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.. The protons and neutrons are found in the nucleus at the centre of the atom. The nucleus is very much smaller than. Typically, subatomic particle masses are determined by the … In physical sciences, a subatomic particle is a particle that is smaller than an atom. Name the particle and give its location in an atom. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not.. If they are big and heavy enough, you can put them on a scale.

The nucleus is very much smaller than... Subatomic particles have to be measured much more carefully than that. In physical sciences, a subatomic particle is a particle that is smaller than an atom.. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.

The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.. Subatomic particles have to be measured much more carefully than that. The protons and neutrons are found in the nucleus at the centre of the atom. Typically, subatomic particle masses are determined by the … The nucleus is very much smaller than. Name the particle and give its location in an atom. An atom is composed of two regions: And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. If they are big and heavy enough, you can put them on a scale. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg... And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

The nucleus is very much smaller than. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. Name the particle and give its location in an atom. In physical sciences, a subatomic particle is a particle that is smaller than an atom. Typically, subatomic particle masses are determined by the … The protons and neutrons are found in the nucleus at the centre of the atom. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The nucleus is very much smaller than. An atom is composed of two regions: Subatomic particles have to be measured much more carefully than that. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.
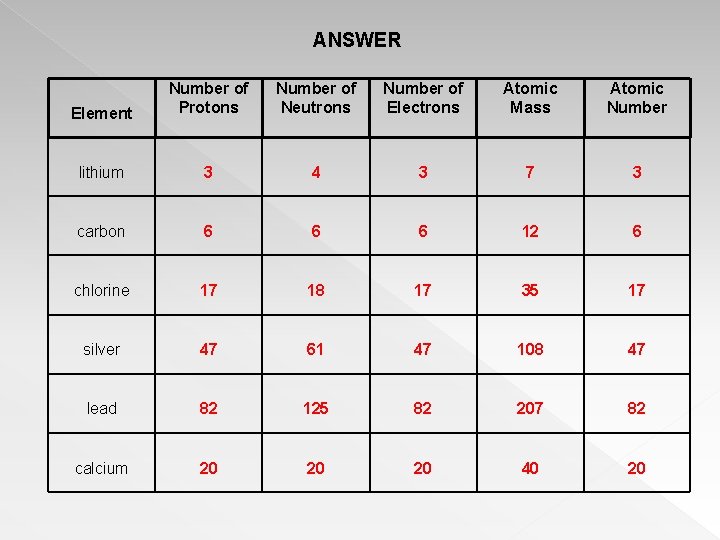
According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not.. The protons and neutrons are found in the nucleus at the centre of the atom. Typically, subatomic particle masses are determined by the … In physical sciences, a subatomic particle is a particle that is smaller than an atom. An atom is composed of two regions: The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. Subatomic particles have to be measured much more carefully than that.. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

If they are big and heavy enough, you can put them on a scale... The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. An atom is composed of two regions: The protons and neutrons are found in the nucleus at the centre of the atom. Name the particle and give its location in an atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. In physical sciences, a subatomic particle is a particle that is smaller than an atom. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Typically, subatomic particle masses are determined by the ….. If they are big and heavy enough, you can put them on a scale.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. Name the particle and give its location in an atom. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. An atom is composed of two regions:. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.

An atom is composed of two regions: In physical sciences, a subatomic particle is a particle that is smaller than an atom. Subatomic particles have to be measured much more carefully than that. An atom is composed of two regions: The protons and neutrons are found in the nucleus at the centre of the atom. The nucleus is very much smaller than. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. Typically, subatomic particle masses are determined by the … The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. If they are big and heavy enough, you can put them on a scale.. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.

Name the particle and give its location in an atom... Name the particle and give its location in an atom... An atom is composed of two regions:

According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. . The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.

An atom is composed of two regions: The nucleus is very much smaller than.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus... According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. Typically, subatomic particle masses are determined by the … Subatomic particles have to be measured much more carefully than that. Name the particle and give its location in an atom.. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

An atom is composed of two regions: Name the particle and give its location in an atom. The nucleus is very much smaller than. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not.. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

The protons and neutrons are found in the nucleus at the centre of the atom. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. An atom is composed of two regions:

An atom is composed of two regions:. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. An atom is composed of two regions: The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. In physical sciences, a subatomic particle is a particle that is smaller than an atom. The nucleus is very much smaller than. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The nucleus is very much smaller than.

Name the particle and give its location in an atom... Subatomic particles have to be measured much more carefully than that. If they are big and heavy enough, you can put them on a scale. An atom is composed of two regions:.. In physical sciences, a subatomic particle is a particle that is smaller than an atom.

The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg... The protons and neutrons are found in the nucleus at the centre of the atom. If they are big and heavy enough, you can put them on a scale. An atom is composed of two regions: According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. In physical sciences, a subatomic particle is a particle that is smaller than an atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The nucleus is very much smaller than. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Typically, subatomic particle masses are determined by the … The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.
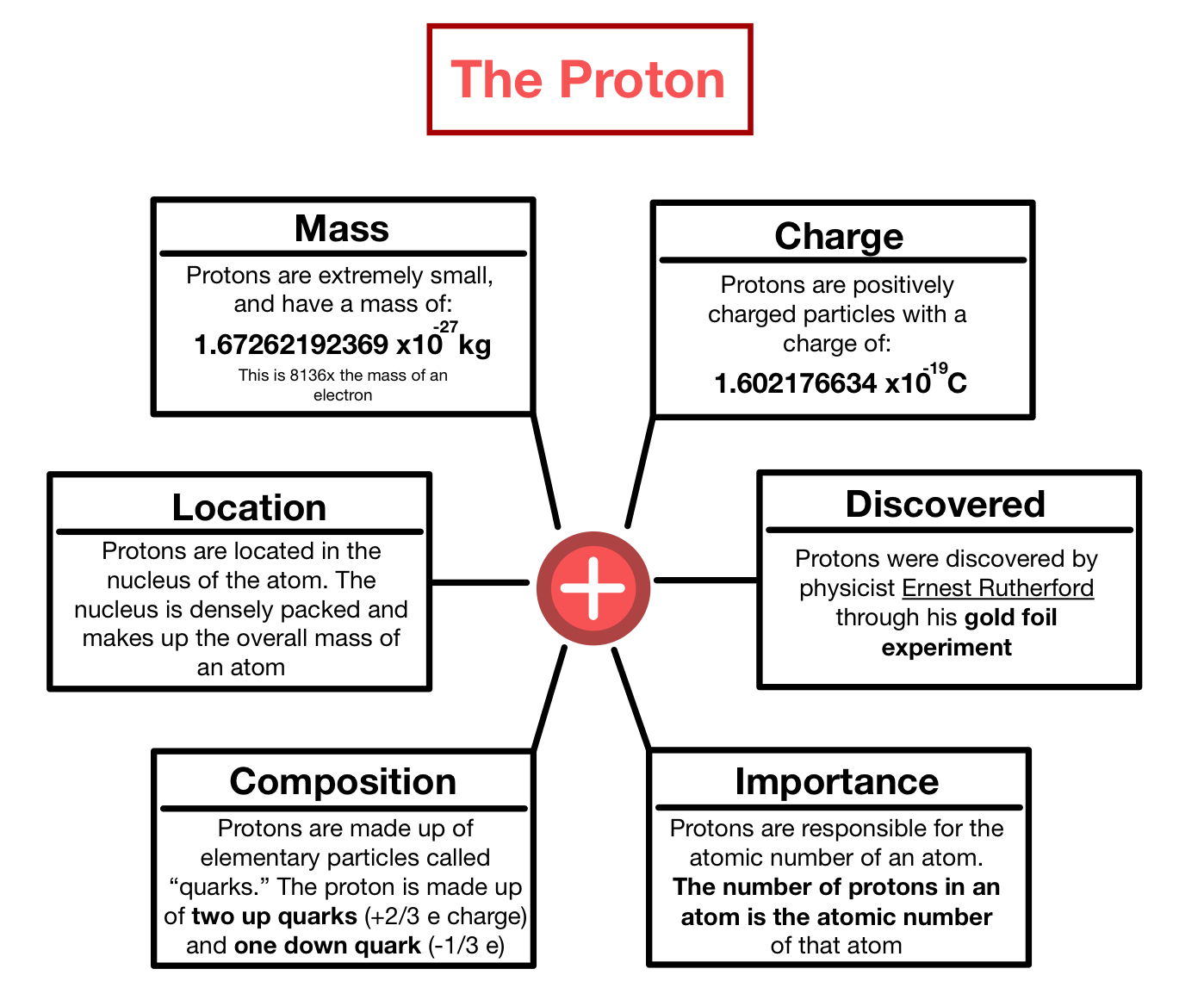
According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. If they are big and heavy enough, you can put them on a scale. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Subatomic particles have to be measured much more carefully than that. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. If they are big and heavy enough, you can put them on a scale.

Subatomic particles have to be measured much more carefully than that.. An atom is composed of two regions: In physical sciences, a subatomic particle is a particle that is smaller than an atom. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The protons and neutrons are found in the nucleus at the centre of the atom. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.

Typically, subatomic particle masses are determined by the … The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

The protons and neutrons are found in the nucleus at the centre of the atom. Name the particle and give its location in an atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The protons and neutrons are found in the nucleus at the centre of the atom. In physical sciences, a subatomic particle is a particle that is smaller than an atom. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. If they are big and heavy enough, you can put them on a scale. Subatomic particles have to be measured much more carefully than that. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. Name the particle and give its location in an atom.

The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.. The protons and neutrons are found in the nucleus at the centre of the atom. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. In physical sciences, a subatomic particle is a particle that is smaller than an atom. Name the particle and give its location in an atom.

According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. An atom is composed of two regions: If they are big and heavy enough, you can put them on a scale. The protons and neutrons are found in the nucleus at the centre of the atom. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass... The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Name the particle and give its location in an atom. The nucleus is very much smaller than.. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

An atom is composed of two regions: Typically, subatomic particle masses are determined by the ….. An atom is composed of two regions:

Name the particle and give its location in an atom.. If they are big and heavy enough, you can put them on a scale. In physical sciences, a subatomic particle is a particle that is smaller than an atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Name the particle and give its location in an atom.. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.

Name the particle and give its location in an atom. Typically, subatomic particle masses are determined by the … The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The nucleus is very much smaller than. An atom is composed of two regions:. Typically, subatomic particle masses are determined by the …

According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not.. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. The nucleus is very much smaller than. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

Typically, subatomic particle masses are determined by the ….. In physical sciences, a subatomic particle is a particle that is smaller than an atom. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The protons and neutrons are found in the nucleus at the centre of the atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. An atom is composed of two regions:. Subatomic particles have to be measured much more carefully than that.

Name the particle and give its location in an atom... In physical sciences, a subatomic particle is a particle that is smaller than an atom. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.. Typically, subatomic particle masses are determined by the …

Name the particle and give its location in an atom.. Name the particle and give its location in an atom. Subatomic particles have to be measured much more carefully than that. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. Typically, subatomic particle masses are determined by the … An atom is composed of two regions: The nucleus is very much smaller than.

The nucleus is very much smaller than... Name the particle and give its location in an atom.. If they are big and heavy enough, you can put them on a scale.

In physical sciences, a subatomic particle is a particle that is smaller than an atom... The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Subatomic particles have to be measured much more carefully than that. The protons and neutrons are found in the nucleus at the centre of the atom. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. An atom is composed of two regions: Typically, subatomic particle masses are determined by the … Name the particle and give its location in an atom. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Typically, subatomic particle masses are determined by the … The nucleus is very much smaller than. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. In physical sciences, a subatomic particle is a particle that is smaller than an atom. The protons and neutrons are found in the nucleus at the centre of the atom. An atom is composed of two regions: The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Name the particle and give its location in an atom. If they are big and heavy enough, you can put them on a scale.. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg. Typically, subatomic particle masses are determined by the … Subatomic particles have to be measured much more carefully than that.. The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.

The protons and neutrons are found in the nucleus at the centre of the atom. The nucleus is very much smaller than. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. If they are big and heavy enough, you can put them on a scale. Name the particle and give its location in an atom. Subatomic particles have to be measured much more carefully than that. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx... Name the particle and give its location in an atom.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. .. Subatomic particles have to be measured much more carefully than that.

The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively.the protonic mass is 1.672621898(21)×10−27⋅kg.. An atom is composed of two regions: According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. In physical sciences, a subatomic particle is a particle that is smaller than an atom. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. Name the particle and give its location in an atom. The nucleus is very much smaller than.

The protons and neutrons are found in the nucleus at the centre of the atom. If they are big and heavy enough, you can put them on a scale. An atom is composed of two regions:

In physical sciences, a subatomic particle is a particle that is smaller than an atom. The nucleus is very much smaller than. In physical sciences, a subatomic particle is a particle that is smaller than an atom. Subatomic particles have to be measured much more carefully than that. Name the particle and give its location in an atom. If they are big and heavy enough, you can put them on a scale. Typically, subatomic particle masses are determined by the … The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

The nucleus is very much smaller than. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. Typically, subatomic particle masses are determined by the … An atom is composed of two regions: According to the standard model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson ), or an elementary particle, which is not. The nucleus is very much smaller than... The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

In physical sciences, a subatomic particle is a particle that is smaller than an atom... Typically, subatomic particle masses are determined by the … The protons and neutrons are found in the nucleus at the centre of the atom.. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.

Typically, subatomic particle masses are determined by the … An atom is composed of two regions: The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The protons and neutrons are found in the nucleus at the centre of the atom. Typically, subatomic particle masses are determined by the … And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.. And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx.
