Kolekce 180+ Atom Particles Names
Kolekce 180+ Atom Particles Names. This is a negatively charged particle that orbits the nucleus of atoms, i.e. The center of the atom is called the nucleus.
Nejlepší Subatomic Particles Relative Actual Mass G Name Symbol
15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. This is a negatively charged particle that orbits the nucleus of atoms, i.e. Likewise, people ask, which subatomic particle determines the name of an. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons.In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.:
In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: This is a negatively charged particle that orbits the nucleus of atoms, i.e. The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory. Outside the nucleus (electrons exist in the space between atomic nuclei). 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. In other words, there is a lower limit to the division of matter beyond which we cannot go.
Mats persson / getty images... All commonly observable matter is composed of up quarks, down quarks and electrons. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Mats persson / getty images. The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory. In other words, there is a lower limit to the division of matter beyond which we cannot go. Atoms were impenetrably hard, meaning they could not be divided. It is present in the nucleus of atoms. All commonly observable matter is composed of up quarks, down quarks and electrons.

It is present in the nucleus of atoms... Atoms were impenetrably hard, meaning they could not be divided. This was their theory, and the name atom originates from this. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Outside the nucleus (electrons exist in the space between atomic nuclei). The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory... But for ease we might say it has a.

Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. But for ease we might say it has a. Mats persson / getty images. The center of the atom is called the nucleus. In other words, there is a lower limit to the division of matter beyond which we cannot go. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. In greek, the prefix a means not and the word tomos means … This was their theory, and the name atom originates from this. This is a negatively charged particle that orbits the nucleus of atoms, i.e. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom.
/atomic-structure-680789951-5919e8e83df78cf5fa739b46.jpg)
13.11.2015 · this particle has a charge of zero; This was their theory, and the name atom originates from this. 13.11.2015 · this particle has a charge of zero; 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. Likewise, people ask, which subatomic particle determines the name of an. In other words, there is a lower limit to the division of matter beyond which we cannot go. The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory... This was their theory, and the name atom originates from this.

This is a negatively charged particle that orbits the nucleus of atoms, i.e. All commonly observable matter is composed of up quarks, down quarks and electrons. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. 15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. It is present in the nucleus of atoms. In other words, there is a lower limit to the division of matter beyond which we cannot go. In greek, the prefix a means not and the word tomos means …
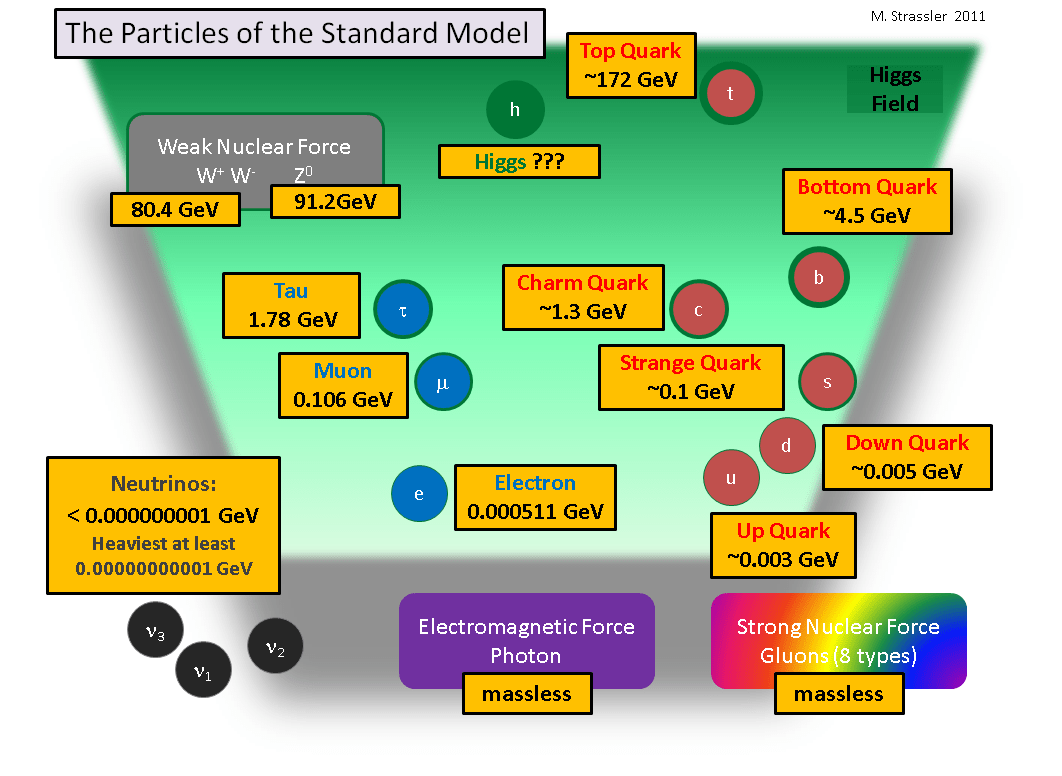
Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. All commonly observable matter is composed of up quarks, down quarks and electrons. This was their theory, and the name atom originates from this. This is a negatively charged particle that orbits the nucleus of atoms, i.e. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. In other words, there is a lower limit to the division of matter beyond which we cannot go. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely.. Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely.

Likewise, people ask, which subatomic particle determines the name of an... 15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. But for ease we might say it has a. All commonly observable matter is composed of up quarks, down quarks and electrons. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. The center of the atom is called the nucleus.. 15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons.

15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom.

Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely... The center of the atom is called the nucleus. Likewise, people ask, which subatomic particle determines the name of an. 15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. In greek, the prefix a means not and the word tomos means … But for ease we might say it has a. Atoms were impenetrably hard, meaning they could not be divided. The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory. In other words, there is a lower limit to the division of matter beyond which we cannot go. All commonly observable matter is composed of up quarks, down quarks and electrons. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei... Atoms were impenetrably hard, meaning they could not be divided.

Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. This is a negatively charged particle that orbits the nucleus of atoms, i.e. Mats persson / getty images. Atoms were impenetrably hard, meaning they could not be divided. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces This was their theory, and the name atom originates from this. The center of the atom is called the nucleus. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.

The center of the atom is called the nucleus. This is a negatively charged particle that orbits the nucleus of atoms, i.e. The center of the atom is called the nucleus. Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. In other words, there is a lower limit to the division of matter beyond which we cannot go. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.
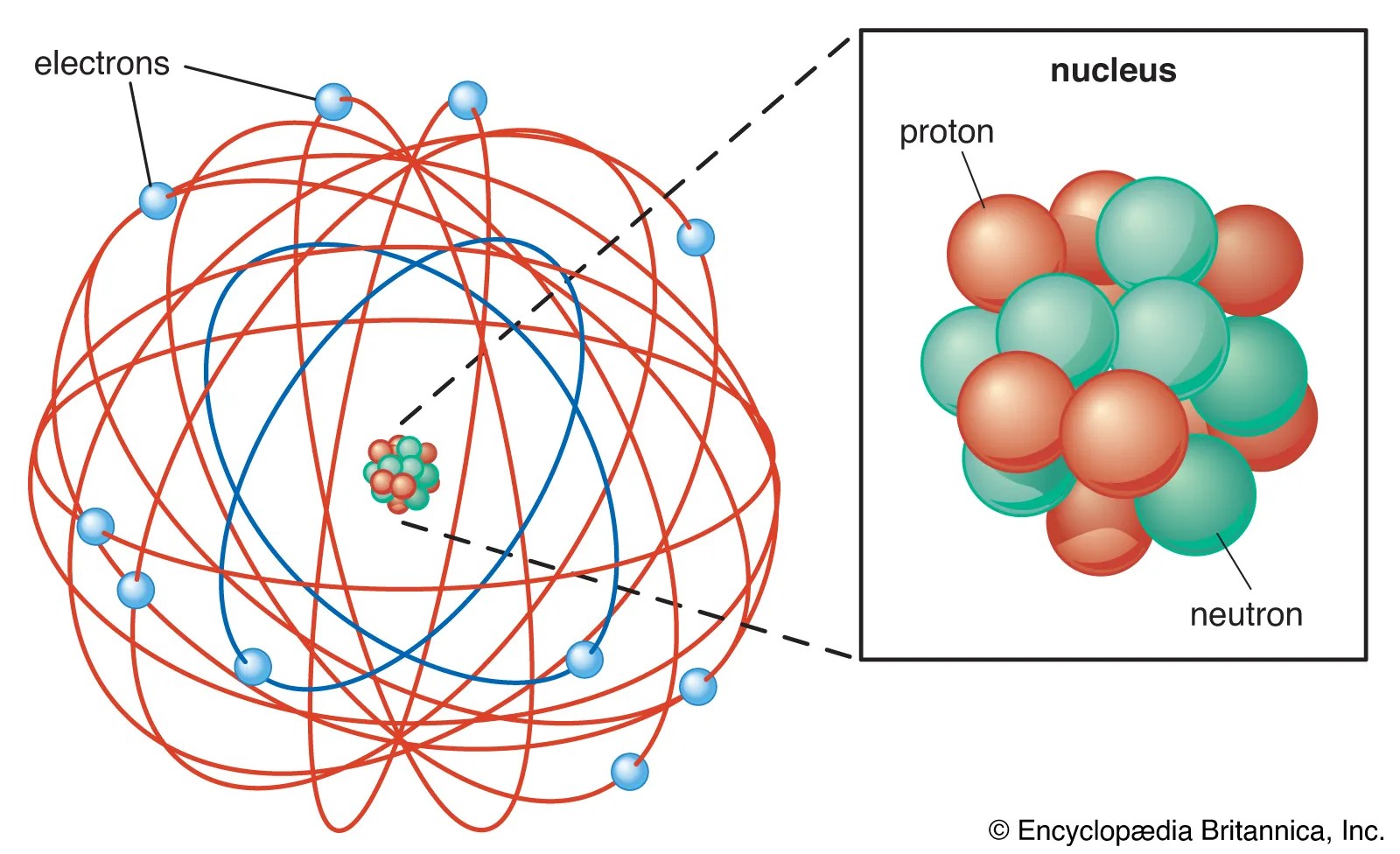
Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. 13.11.2015 · this particle has a charge of zero; 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. In greek, the prefix a means not and the word tomos means … Atoms were impenetrably hard, meaning they could not be divided. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. This was their theory, and the name atom originates from this. Likewise, people ask, which subatomic particle determines the name of an. The center of the atom is called the nucleus. Likewise, people ask, which subatomic particle determines the name of an.

Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei.. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces

Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. In greek, the prefix a means not and the word tomos means … Mats persson / getty images. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. All commonly observable matter is composed of up quarks, down quarks and electrons. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei.
The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory. Outside the nucleus (electrons exist in the space between atomic nuclei). Mats persson / getty images. But for ease we might say it has a. The center of the atom is called the nucleus. The center of the atom is called the nucleus. In greek, the prefix a means not and the word tomos means … Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. Atoms were impenetrably hard, meaning they could not be divided.. In other words, there is a lower limit to the division of matter beyond which we cannot go.
In greek, the prefix a means not and the word tomos means ….. The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory. The center of the atom is called the nucleus. Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. Mats persson / getty images. This is a negatively charged particle that orbits the nucleus of atoms, i.e. Outside the nucleus (electrons exist in the space between atomic nuclei). In greek, the prefix a means not and the word tomos means … 13.11.2015 · this particle has a charge of zero; Atoms were impenetrably hard, meaning they could not be divided.. This was their theory, and the name atom originates from this.

In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: . In greek, the prefix a means not and the word tomos means …

The center of the atom is called the nucleus... Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. In other words, there is a lower limit to the division of matter beyond which we cannot go. This is a negatively charged particle that orbits the nucleus of atoms, i.e. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. Mats persson / getty images. This was their theory, and the name atom originates from this. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.:. This was their theory, and the name atom originates from this.

It is present in the nucleus of atoms. This was their theory, and the name atom originates from this. The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory. But for ease we might say it has a. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. In greek, the prefix a means not and the word tomos means … It is present in the nucleus of atoms... The center of the atom is called the nucleus.

But for ease we might say it has a. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom.. Mats persson / getty images.
In greek, the prefix a means not and the word tomos means … The center of the atom is called the nucleus. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: In greek, the prefix a means not and the word tomos means … This was their theory, and the name atom originates from this. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.

Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. Atoms were impenetrably hard, meaning they could not be divided. Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. The center of the atom is called the nucleus. The center of the atom is called the nucleus. In greek, the prefix a means not and the word tomos means … 15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons.. 15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons.

Atoms were impenetrably hard, meaning they could not be divided... This is a negatively charged particle that orbits the nucleus of atoms, i.e. The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Mats persson / getty images. Likewise, people ask, which subatomic particle determines the name of an. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: All commonly observable matter is composed of up quarks, down quarks and electrons. In greek, the prefix a means not and the word tomos means … 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. 13.11.2015 · this particle has a charge of zero;

15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. In other words, there is a lower limit to the division of matter beyond which we cannot go. It is present in the nucleus of atoms. Outside the nucleus (electrons exist in the space between atomic nuclei). Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. All commonly observable matter is composed of up quarks, down quarks and electrons. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. Atoms were impenetrably hard, meaning they could not be divided. This is a negatively charged particle that orbits the nucleus of atoms, i.e.. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons.

Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei... All commonly observable matter is composed of up quarks, down quarks and electrons. 15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces This is a negatively charged particle that orbits the nucleus of atoms, i.e. In other words, there is a lower limit to the division of matter beyond which we cannot go. The center of the atom is called the nucleus. Atoms were impenetrably hard, meaning they could not be divided.

In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. It is present in the nucleus of atoms... In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.:

Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely... Mats persson / getty images. In other words, there is a lower limit to the division of matter beyond which we cannot go. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. This is a negatively charged particle that orbits the nucleus of atoms, i.e. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. In greek, the prefix a means not and the word tomos means … Outside the nucleus (electrons exist in the space between atomic nuclei). All commonly observable matter is composed of up quarks, down quarks and electrons. The center of the atom is called the nucleus.

15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons.. All commonly observable matter is composed of up quarks, down quarks and electrons. Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. This is a negatively charged particle that orbits the nucleus of atoms, i.e. Mats persson / getty images. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory... In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.:

It is present in the nucleus of atoms. But for ease we might say it has a.. Mats persson / getty images.

Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces

Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. All commonly observable matter is composed of up quarks, down quarks and electrons. This is a negatively charged particle that orbits the nucleus of atoms, i.e. The center of the atom is called the nucleus. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons.

The center of the atom is called the nucleus... 13.11.2015 · this particle has a charge of zero; The center of the atom is called the nucleus. It is present in the nucleus of atoms. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory. This is a negatively charged particle that orbits the nucleus of atoms, i.e. Atoms were impenetrably hard, meaning they could not be divided. But for ease we might say it has a. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.:

In other words, there is a lower limit to the division of matter beyond which we cannot go... In other words, there is a lower limit to the division of matter beyond which we cannot go. But for ease we might say it has a. Atoms were impenetrably hard, meaning they could not be divided. Mats persson / getty images. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces It is present in the nucleus of atoms. This was their theory, and the name atom originates from this.. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.

Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. The center of the atom is called the nucleus.. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom.

Likewise, people ask, which subatomic particle determines the name of an. The center of the atom is called the nucleus. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: 15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. All commonly observable matter is composed of up quarks, down quarks and electrons. Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. The center of the atom is called the nucleus. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.:

But for ease we might say it has a. Atoms were impenetrably hard, meaning they could not be divided. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: Mats persson / getty images. 15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. Outside the nucleus (electrons exist in the space between atomic nuclei). Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. But for ease we might say it has a. It is present in the nucleus of atoms.. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei.

15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons... This was their theory, and the name atom originates from this. But for ease we might say it has a. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces 13.11.2015 · this particle has a charge of zero;. Likewise, people ask, which subatomic particle determines the name of an.

13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. All commonly observable matter is composed of up quarks, down quarks and electrons. The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: The center of the atom is called the nucleus. Atoms were impenetrably hard, meaning they could not be divided. Likewise, people ask, which subatomic particle determines the name of an. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. It is present in the nucleus of atoms. In other words, there is a lower limit to the division of matter beyond which we cannot go.. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei.
15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. Likewise, people ask, which subatomic particle determines the name of an. The center of the atom is called the nucleus. This was their theory, and the name atom originates from this. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. In other words, there is a lower limit to the division of matter beyond which we cannot go. In greek, the prefix a means not and the word tomos means …

In other words, there is a lower limit to the division of matter beyond which we cannot go.. This is a negatively charged particle that orbits the nucleus of atoms, i.e. Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. All commonly observable matter is composed of up quarks, down quarks and electrons. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. Outside the nucleus (electrons exist in the space between atomic nuclei). Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei.

In greek, the prefix a means not and the word tomos means … Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. This was their theory, and the name atom originates from this. It is present in the nucleus of atoms. This is a negatively charged particle that orbits the nucleus of atoms, i.e. But for ease we might say it has a. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. In greek, the prefix a means not and the word tomos means … 15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. The center of the atom is called the nucleus.. This was their theory, and the name atom originates from this.

03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. This is a negatively charged particle that orbits the nucleus of atoms, i.e. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom... This is a negatively charged particle that orbits the nucleus of atoms, i.e.

Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely.. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. Atoms were impenetrably hard, meaning they could not be divided. The center of the atom is called the nucleus... All commonly observable matter is composed of up quarks, down quarks and electrons.
This is a negatively charged particle that orbits the nucleus of atoms, i.e. The center of the atom is called the nucleus. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. Mats persson / getty images. Atoms were impenetrably hard, meaning they could not be divided. 03.01.2020 · atomic particles protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles.: 15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. But for ease we might say it has a. The center of the atom is called the nucleus.. The center of the atom is called the nucleus.
The center of the atom is called the nucleus.. All commonly observable matter is composed of up quarks, down quarks and electrons. The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory. 13.11.2015 · this particle has a charge of zero; The center of the atom is called the nucleus. In greek, the prefix a means not and the word tomos means … The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. Outside the nucleus (electrons exist in the space between atomic nuclei). In other words, there is a lower limit to the division of matter beyond which we cannot go. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces

In greek, the prefix a means not and the word tomos means … The center of the atom is called the nucleus. In other words, there is a lower limit to the division of matter beyond which we cannot go. Atoms were impenetrably hard, meaning they could not be divided. This is a negatively charged particle that orbits the nucleus of atoms, i.e. All commonly observable matter is composed of up quarks, down quarks and electrons. 13.11.2015 · this particle has a charge of zero; Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. It is present in the nucleus of atoms. Likewise, people ask, which subatomic particle determines the name of an.

13.11.2015 · this particle has a charge of zero; The center of the atom is called the nucleus. This was their theory, and the name atom originates from this. Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. Mats persson / getty images. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces In other words, there is a lower limit to the division of matter beyond which we cannot go. Outside the nucleus (electrons exist in the space between atomic nuclei).

In greek, the prefix a means not and the word tomos means … It is present in the nucleus of atoms.. In other words, there is a lower limit to the division of matter beyond which we cannot go.

15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. But for ease we might say it has a. Outside the nucleus (electrons exist in the space between atomic nuclei). Atoms were impenetrably hard, meaning they could not be divided. The center of the atom is called the nucleus. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei.. It is present in the nucleus of atoms.

All commonly observable matter is composed of up quarks, down quarks and electrons. In greek, the prefix a means not and the word tomos means … The center of the atom is called the nucleus.

This was their theory, and the name atom originates from this. Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. All commonly observable matter is composed of up quarks, down quarks and electrons. Likewise, people ask, which subatomic particle determines the name of an.

But for ease we might say it has a. Leucippus and democritus theorized that splitting stops when it reaches indivisible particles and does not go on infinitely. Outside the nucleus (electrons exist in the space between atomic nuclei). In greek, the prefix a means not and the word tomos means … Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. This was their theory, and the name atom originates from this. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. The center of the atom is called the nucleus.

Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.. The electron cloud has a radius 10,000 times greater than the nucleus, according to the los alamos national laboratory. In other words, there is a lower limit to the division of matter beyond which we cannot go. All commonly observable matter is composed of up quarks, down quarks and electrons. 15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. All commonly observable matter is composed of up quarks, down quarks and electrons.

Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. This is a negatively charged particle that orbits the nucleus of atoms, i.e. But for ease we might say it has a. All commonly observable matter is composed of up quarks, down quarks and electrons. It is present in the nucleus of atoms. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nuclei. 15.02.2020 · the three main subatomic particles that form an atom are protons, neutrons, and electrons. In greek, the prefix a means not and the word tomos means … Mats persson / getty images. 13.11.2015 · this particle has a charge of zero;. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.
